When it comes to making management decisions for any crop field, such as walnut and almond orchards, it should be a standard practice for growers to collect soil samples to determine whether they have the presence of plant pathogens or key plant parasitic nematodes. In California, plant parasitic nematodes can significantly impact crop production. They are destructive pests that cause considerable damage to plant roots, thus disrupting the uptake of water and nutrients which results in reduced crop vigor and yield.
Plant parasitic nematodes are microscopic roundworms that feed on plant tissues. In walnut orchards, root lesion nematode is a key pest that causes substantial damage and is estimated to be present in roughly 85% of walnut fields. This nematode can negatively impact growth in young trees by invading and destroying smaller root mass. In mature, older trees with more established root systems, the impact is less significant. Nematodes are defined by their feeding lifestyle; root lesion is classified as a migratory endoparasite.
All stages, both juvenile and adult, enter the roots and feed internally, causing damage to plant cells and leaving behind dark necrotic root lesions. Ring nematode, another key pest of walnut, is classified as migratory ectoparasitic. Juveniles and adults feed on the outside of the roots, causing damage and stunting. The spectrum of damage includes lack of vigor, stunting, general decline in growth and lower yield. Wounds inflicted on roots by plant parasitic nematodes can indirectly lead to additional damage by providing an entry point for secondary plant pathogens, such as bacteria/fungi, to invade and promote root rot.
The nematode density and the damage they cause will continue to increase if sufficient susceptible plants are available and favorable environmental conditions persist. There are other nematodes present in walnut that are known to cause damage, but the most significant destruction is done by root lesion.
Diagnosing Nematode Symptoms
Diagnosing crop decline due to nematodes can be quite difficult because very few plant parasitic nematodes cause distinctive diagnostic symptoms. Symptoms can differ greatly and often appear nonspecific. A thorough field diagnosis to determine the presence of plant parasitic nematodes should consider these key factors: foliar and root symptoms, field history and laboratory analysis. Above ground symptoms are generally nondescript and often misdiagnosed as nutrient deficiency or drought stress.
As a grower, when examining your orchard, visually observe differences in growth, including stunting, chlorosis, premature wilting and non-uniform distribution within a field. This will indicate a potential issue, warranting further investigation. You will then want to examine tree roots for symptoms, which is imperative when identifying a nematode problem. Nematode damage on roots is often associated with specific plant parasitic nematode feeding lifestyle. For example, root knot nematode causes galling/swelling knots on roots, whereas root lesion nematode causes lesions/necrosis. The feeding by these nematodes and others causes stunting of the root system and excessive lateral growth.
Another key component in determining nematode diagnosis is examining the field history. This can aid in identifying the presence of plant parasitic nematodes and other pathogen pests. Nematodes present in the field in recent years would have resulted in prior crop decline and are more than likely to be present or will resurface in future seasons.
Sample Soils
Once all of these key factors are evaluated, the most vital component that growers should implement in their program is collecting soil and root samples for laboratory diagnostic analysis. Sending composite soil samples to a lab will confirm the presence of key plant parasitic nematodes and their relative pressure within the field. Knowing the identity of the plant parasitic nematodes and the relative density estimates is critical in determining the right management decisions and pre-treatment measures, such as crop rotation, fumigation and rootstock.
Nematode soil sampling is vital when preparing to plant or replant an orchard. For predictive sampling, soil should be taken prior to planting or preferably at the end of the previous crop/orchard. It is best to collect both roots and soil as nematodes are soilborne pathogens that feed directly and within the roots. The number of plant parasitic nematodes of specific genera can give some indication about the damage potential. When laboratory analysis indicates presence of nematode pressure of key plant parasitic nematodes such as root lesion, ring, or root knot, specific treatment plans can be formulated to target these specific genera.
Treatment and Control
For pre-treatment control, especially with regards to walnut and almond replants, soil fumigation is the standard management tool for growers. The objective is to reduce nematode populations below a threshold to avoid serious crop damage. Even when laboratory tests detect low nematode pressure, the pests are likely to increase in density in the first few years of a new planting. Soil fumigation helps to suppress a broad spectrum of soilborne pests including root rot, prunus replant disorder and plant parasitic nematodes. Mitigating the damage caused by these pests ensures a strong start to the seedlings or dormant propagative material. Fumigation promotes healthy soil for the grower’s long-term investment. This pre-plant measure helps flourishing root systems, which enhances trunk, shoot and canopy growth. There are two fumigants that are commonly applied: Telone II (1,3-dichloropropene), the industry standard pre-plant nematicide, and Chloropicrin, the industry standard pre-plant fungicide. These products can be co-applied or stand alone.
TriCal has been providing pre-plant soil fumigation support to growers over 50 years. Field trial research has demonstrated that orchards starting out in fumigated soil experience rapid seedling establishment, increased initial growth, earlier and larger harvest yields and increased orchard longevity. To determine the right fumigation prescription for an orchard, the rate and method are based on the potential crop and pathogen pressure.
Proper soil preparation and application are necessary for optimal fumigation efficacy. Field preparation should include a deep rip of the field soil and removal of as much old root mass from the prior crop/orchard as possible. This is important because old roots can harbor nematodes such as root lesion and root knot, which can remain dormant for several years after a mature tree has been removed. Heavier textured soils such as clay loam also can produce large clods after tilling, where clods can hold moisture as well as viable nematodes. Clod reduction is important.
At TriCal Diagnostics and TriCal, we help growers with their management decisions by following integrated pest management practices that include pre-plant measures, fumigation and post-plant treatments. TriCal Diagnostics offers disease and plant problem identification services for growers, field personnel and PCAs while conducting problem-solving in the field as well as specialized lab-based research projects. Our primary goal is providing growers guidance and support for their management decisions and help diagnosing plant/nematode issues.
One of our main foci in TriCal R&D is conducting field trial research. We have initiated field trials throughout the Valley and Northern California, on various key crops to examine the performance of fumigant treatments using Telone II and Chloropicrin. We are actively investigating different application depths, rates of both fumigants in combination or stand alone and IPM nematode management programs (e.g. pre-plant fumigation followed by post-plant contact nematicide). Nematode control within a field requires integration of multiple strategies to provide effective, long-term crop protection.

Fumigant Research
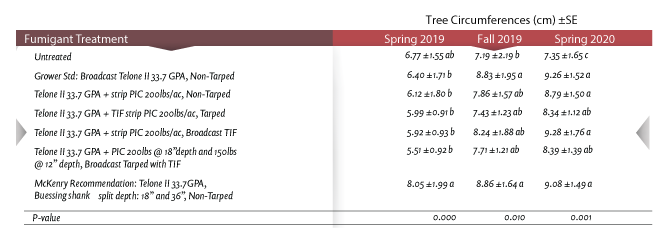
A multi-year walnut replant demo trial was established in Fall 2018 in Yolo County to evaluate six different one-acre fumigant treatments versus an untreated area within an 84-acre field. The primary objective of the trial was to examine performance of the grower standard (Telone II at max rate of 33.5 GPA with an 18-inch injection depth) compared to other fumigant treatments, which included variations of TriCal’s go-to treatment for replant disorder (Telone II with strip or broadcast Chloropicrin, tarped and non-tarped treatment variations) and the recommendation from Dr. McKenry (UC emeritus), which relies on Telone II applied by the Buessing shank (split fumigant injection depth at 18 and 36 inches) in suppressing root lesion nematode populations. We conducted thorough evaluations each season by identifying key plant parasitic nematodes and determining relative pressure post-fumigation every fall and spring as well as collecting tree growth metrics. Pre-treatment nematode assays revealed significant pressure from lesion nematode (average populations of 200 to 350 nematodes/250 g soil), where the economic threshold for lesion nematode is often cited as being 1 nematode/250 g soil. Soil fumigation occurred in Fall 2018. Initial post-fumigation nematode evaluation was done in Spring 2019. No plant parasitic nematodes were detected within the fumigant treatments, while significant root lesion nematode density was evident within the untreated area of the field. Through monitoring and sampling, we documented sustained nematode suppression through 2020. Treatment areas continue to demonstrate a significant reduction in root lesion pressure compared to the untreated control.
Tree circumference over time is a measurable indicator of the benefits of fumigation. In Spring 2020, all fumigant treatments demonstrated significantly greater tree growth compared to the untreated control (One-Way ANOVA, P-value 0.001, Table 1). Trees on untreated ground were smaller in size and had less canopy growth than did the trees within the fumigant treatment. On average and over the 2020 year, the fumigant treatments resulted in a 45% to 55% increase in tree growth from initial Spring 2019 measurements compared to untreated control, which only exhibited an 8% increase over the same period. Monitoring the fumigant treatment blocks and commercial field through the seasons has shown the benefits fumigated soil can provide when establishing a young orchard. This is critical to formulate management decisions for orchard longevity. A key takeaway is to highlight the importance of pre-fumigation soil sampling to determine the appropriate pre-treatment pest control prescription for when starting a new orchard. Additionally, soil sampling for nematode post-fumigation and post-planting should be done to monitor the duration of nematode suppression offered by Telone II (with or without Chloropicrin) and to assess the possible future need for integrating post-plant contact nematicides into your agronomic program once the initial (multi-year) benefits of fumigation taper off.